
ARCHIVE FILE
This article was published in 1986
See the original document
The control of gastro-intestinal parasites of goats
Betty Hall, Veterinary Officer, Wormkill Co-ordinator N.S.W. Department of Agriculture, Armidale
Summary
The problems inherent in controlling gastro-intestinal parasites in goats are reviewed with particular reference to the use of the Wormkill strategic programme which is designed to control H. contortus and T. colubriformis in the summer rainfall area of the Northern Tablelands of N.S.W.
The removal of caprines from their more natural dry environs with progressive loss of adequate browse and stocking rate intensification, subjects them to an assault from internal parasites for which they are ill-equipped. The loss of hybrid vigour as ferals are up-graded towards the mohair or cashmere purebreds aggravates this situation.
Control measures are eroded by certain management practices which increase stocking rates, at critical times. To forestall loss of kids from predators a system of folding is instituted which can mean the concentration of does and kids for up to six weeks in a small 'safe' area. This kidding area is often used as a weaning paddock because of its security and shelter advantages.
These practices subject does and kids at their most susceptible times to high stocking rates and high pasture contamination levels.
These intensively used areas are utilised year after year.
A review of the literature and practical experience indicate that:
1. Goats are more susceptible to the effects of internal parasites than sheep and recovery may be more protracted.
2. Goats may have evolved a less efficient age-acquired resistance than sheep because of their natural browsing habits.
Goats are far more susceptible to stocking rate effects than sheep and will carry higher burdens than sheep run at the same stocking rate.
4. There is a great deal of speculation surrounding the use of anthelmintics in goats, the correct dose rates and the metabolism of anthelmintics. Common practice among goat producers has been to administer anthelmintics at cattle dose rate levels, better results ensuing from this procedure.
Certainly anthelmintic resistance or ineffectivity has to be watched closely.
5. The practices which accelerate the development of anthelmintic resistance have to be continually re-addressed. Where of course one is forced to use anthelmintics which are not registered for use in goats or are recommended for use at dose rates which apply to sheep and therefore may not be appropriate, one has to be prepared for less than acceptable results.
Despite all these factors, control of internal parasites in goats is attainable and graziers appreciate an honest approach where all known and unknown components are explained.
Introduction
The expansion of the mohair and cashmere industries is dependent on the acceptance of the goat into the management systems of the sheep grazing industry. Diversification is being encouraged both by the proponents of the angora or cashmere goat and by the Department of Agriculture. Areas where prime lamb and wool production are viable should be suitable for goat fibre production. Advantages in terms of the complementary grazing habits of goats and sheep or cattle, the valuable by-products and weed control have already encouraged many entrenched sheepmen into importing goats onto properties in the Northern Tablelands and Slopes. Typical orders five years ago were for 30 or 40 animals, recent sales involve flocks of 500 or more.
Many Australian ferals with angora or cashmere traits are being moved from drier inland districts such as Cobar and Bourke, to be used in upgrading programmes in higher rainfall zones. Low grade parasitism is normal in feral goats and presents no hazard to health. The goat's unique selective grazing behaviour, its ability to travel, climb and reach further than other ruminants and its natural tendency to avoid wet areas are protective barriers against internal parasites.
Parasitism becomes an escalating problem as goats are grazed on sown, improved pastures with little or no access to browse and as stocking rates are intensified. Experience has also shown that upgraded goats in particular purebred angoras, are far more susceptible to parasites than their feral counterparts.
The situation on the Northern Tablelands and Slopes is that we have a great variety of situations under which goats are run. From the relatively small stud on 100ha or less to the extensively run near feral goats used to clear gullies and gorges of blackberries and all levels of management and up-grading programmes in between. Often on one property a whole range of management strategies can be experienced whereby parasite control in the extensively run goats is no problem but as the selection process progresses so the grazing management changes. It is at the point where we have up-graded stock depastured on improved country or crop that parasitism hits its peak and control programmes are an essential if disastrous situations are to be avoided.
The launching of the Wormkill programme in July 1984 was perhaps fortuitous for the goat industry (for which it was not designed) and for veterinarians in that region. Goat producers adopted the programme for one of two reasons:
1. They were desperate for advice on parasite control and anything for sheep was seen to be as near as they would get for specific guidelines.
2. Veterinarians advised that goats running with sheep or on the same property as sheep should be put onto the same control programme, for obvious reasons.
We have no statistics for the uptake of Worrkill by the goat industry other than personal communication and feed-back at goat producer field days. This indicates that the majority would be on Wormkill and are delighted with the results in comparison to the haphazard drenching programmes used previously and the excellent results afforded by Wormkill. Producers express the same concerns about the efficiency of Wormkill in young goats as sheepmen do about Wormkill and prime lamb production. In all cases investigated where parasite control has been less than effective, it could be said to be due to:
1. Anthelmintic ineffectivity and/or resistance.
2. Gross under estimation of live weights and subsequent under-dosing.
3. Extremely high stocking rates in combination with marginal or gross anthelmintic resistance.
4. Undernutrition thus increasing the susceptibility of the goats to higher parasite burdens.
The development of internal parasite control programmes for use in the caprine is fraught with many problems and uncertainties which Wormkill has shown are not necessarily insurmountable.
There is an obvious need for ongoing research and investigation in the sphere of anthelmintic resistance, metabolism and dose rates in goats.
The surveys conducted by Kettle et al. in New Zealand showed that resistant strains of nematodes from goats retained their resistant status in sheep. If we are not to compound the already critical situation on the Northern Tablelands with respect to anthelmintic resistance in sheep then we need answers fast and before the goat industry experiences further accelerated expansion.
Designing programmes for use in goats
Many dilemmas face veterinarians when attempting to formulate programmes for internal parasite control in goats.
Their lack of knowledge about the industry, the diversity of management procedures and the insecurity which comes from using anthelmintics not registered for use in goats have to be overcome.
The major quandary is whether or not we can apply the same parasitological principles, known to be effective in sheep, to goats.
Once we have overcome our own natural reluctance to become involved in the industry we can begin the process of educating both producers and ourselves. This process is very rewarding and if our investigations are conducted in a systematic way, with careful monitoring and evaluation of our results we can formulate strategies for control of internal parasites in goats which are not dependent on suppressive drenching regimes.
I did not make a conscious decision to become actively involved in the goat industry or internal parasite control in caprines. The industry took Wormkill to its bosom and I suppose those of us actively involved in the programme were pulled into the industry as a natural consequence.
What I have learned in the past eighteen months does not make me an expert but it has made me more confident in my approach.
I do, however constantly remind myself that if things go wrong, it will be catastrophic and a great deal of effort will be required to correct the situation. Animal husbandry principles have to be applied in the convalescent period and recovery will be protracted. Losses can be expected to be greater than a similar situation in sheep.
I also remind myself of the dangers of extrapolating from findings in dairy goats or angoras and applying the results to all goats.
Review of known factors and grey areas involved in parasitism in goats
1. Pathogens and pasture effects
The main pathogens affecting goats are H. contortus, T. colubriformis and O. circumcincta. Haemonchosis is a particular problem in summer rainfall areas and western districts during wet spells. Winter rainfall areas have only the little 'black and brown fellas' to cope with.
There is evidence to show that whilst sheep and goats share the same parasites there is a marked difference between levels of infestation.
Le Jambre and Royal showed that young sheep and goats up to 7 months of age were equally susceptible to internal parasites. However, angora wethers over 15 months of age carried more parasites than sheep of the same age. Those paddocks grazed only by goats being more heavily contaminated than those grazed by sheep alone.
Conflicting evidence from Tanzania (McCullock and Kasimbala, 1968) and from Cyprus (Le Rich et al. 1973) showed that adult sheep had heavier infestations than goats.
This conflict could be resolved by comparing the grazing strategies involved.
In the Le Jambre/Royal investigations the animals grazed the same sown pastures with no access to browse, whilst in Tanzania and Cyprus the animals grazed dissimilar pasture types over a large area, the goats having access to browse.
If the goat has evolved a less sophisticated or less efficient acquired age resistance than sheep, then producers must be acquainted with the dangers involved in removing this safety mechanism, browse, from the goat's diet. Silangwa and Todd (1964) demonstrated that if goats browse over 12.5cms they will avoid 99% of infective larvae.
Further work by Le Jambre (1984) showed that angoras are more sensitive to stocking rate effects than Merinos and will have higher burdens than sheep grazed at similar stocking rates particularly if they are forced to graze closer and closer to the soil surface.
Work on the Northern Tablelands has confirmed these findings with respect to stocking rates and type of de-pasturing and access to browse. It has also confirmed that goats are more sensitive to the effects of parasites.
The Wormkill programme (Fig. I) relies very heavily on the age acquired resistance which allows adult dry sheep to go for long periods without the necessity to apply broad-spectrum drenches to control particularly T. colubriformis and also O. circumcincta. Ewes are 'cared for' in a parasitological sense during their susceptible period of lactation by giving pre and post lambing broad-spectrum anthelmintics. The post lambing drench being far more important in my opinion.
The indication is that we may not be able to rely on this natural aid to parasite control and pasture contamination in adult dry goats.
Producers using Wormkill in goats are quite confident about its use in their mature, dry goats and are not concerned about the long period of up to 9 months when no broad-spectrum drench is applied.
Certainly our routine monitoring on a limited number of flocks, gives us no cause for concern in this area and compare favourably with the results in sheep.
Pre- 'August' drench monitoring in dry adult goats. This includes yearling goats and in kid does. All results from properties using standard Wormkill.
| Animal group | Period | Mean Strongyl Larval Differentiation % | |||
|---|---|---|---|---|---|
| e.p.g. | H. | T. | Ost. | ||
| 1. Does | Late July | 257 | 1 | 82 | 12 |
| Yearlings | 1985 | 271 | 1 | 82 | 12 |
| 2. Does | Early August 1985 | 260 | - | 60 | 40 |
There will certainly be a number of producers adding an extra broad spectrum drench for adult goats in late autumn. By extrapolating from the survey questionnaire results used in the Northern Tablelands where 46% of producers admitted to giving an additional drench to ewes in the April/May period we might assume that same applies in goat flocks, This is an insurance policy against fears of the consequences of not doing so.
I am still not sure whether or not the success of Wormkill in adult dry goats is due to a reasonable level of resistance to the establishment of parasites and the ability to throw off burdens or due to the different management practices for older goats.
Once kids are weaned the does are run more extensively with perhaps a greater access to browse and a lower stocking rate. Wethers, similarly are run under more suitable and natural conditions. This is not true for the smaller (in terms of hectarage) goat establishments where browse is unavailable and extensive ranging conditions cannot be supplied. Those graziers running goats under these circumstances are still confident that their older goats can and do take care of themselves.
We have therefore to assume that our efforts should be concentrated on highly susceptible stock, the lactating does, suckling and weanling kids up to 12 months of age and the dangers of overstocking and nil access to browse.
Goat producers constantly re-assure me that Wormkill is the best thing that has happened for the goat industry. Surpassing previous practices and reducing incidental animal health problems and maximising production potential.
Failures when recorded are caused primarily by anthelmintic resistance and exacerbated occasionally by under-nutrition and overstocking.
2. Anthelmintics
Whatever other control strategies are employed to minimise infestation with nematode parasites some, if not all, reliance will have to be placed on anthelmintics.
Anthelmintic resistance in both sheep and goats is becoming more prevalent and more cases are being evaluated weekly in the Northern Tablelands. It is a particular problem in H. contortus endemic areas and where climate is ideally suitable for the development and survival of the free - living stages of the parasites on the pasture. These circumstances have of necessity, ensured that drenching is practised extensively and intensively.
The major thrust from veterinarians is, or should be, the advocation of the principles which will delay or minimise the selection for drench resistance. One has to be in the position to supply a programme incorporating such principles.
1. The minimal use of broad-spectrum drenches, applied at strategically important times to minimise pasture contamination.
2. The use of narrow-spectrum drenches where appropriate and
3. The importance of never under-dosing.
Estimating weights correctly.
Dose to the heaviest in the group.
Check the accuracy of the drench gun.
Check the accuracy of the operator and drenching technique.
Reading and interpreting labels on anthelmintics correctly.
Principles 1 and 2 are dependent on the epidemiological characteristics in each climatic and geographic area. It is unfortunate that these have as yet to be defined in coastal and near coastal areas, particularly the North Coast of N.S.W.
Principle 3 is critical and incorporates a major grey area which must be clarified.
As yet those anthelmintics registered for use in goats are, by inference, recommended to be used at sheep dose rates. Many producers have found by trial and error that better results are obtained by using cattle dose rates or at least increased rates compared to those recommended for sheep.
Veterinarians in the Department of Primary Industry in Fiji are faced with horrendous parasite problems in their goat industry. Their recommendations on dose rates for the benzimidazole anthelmintics are 1ml/3.5kg (sheep 1ml/5kg) because of the rapid rate of clearance of these drenches in goats and the suspicion that this is related to dose rate.
My own recommendations tend to be less specific but encourages at least a rate of 50% up on sheep recommended rates for both benzimidazoles and levamisoles.
If sub-optimal doses are used then we may be selecting more rapidly for anthelmintic resistance than if levels were therapeutically correct for goats.
The efficiency of drenches can generally be related to the pattern of blood levels and the duration above a certain level considered critical.
Kettle and White (1982 unpublished), demonstrated that goats metabolise drenches at a higher rate than sheep and suggested that dose rates for goats need revision.
Figures 2 and 3 indicate that with both levamisoles and the benzimidazoles, the area under the plasma concentration curve, is much less in goats than sheep when dosed at the dose rates recommended for sheep.
Gilham and Obendorf (1985) made similar observations to Kettle and White. Strains of T. colubriformis were confirmed to be resistant to the effects of benzimidazoles, morantel and levamisoles in goats. However, in sheep, the same strain was susceptible to levamisoles. The different therapeutic response in sheep and goats suggested that the pharmacodynamics of levamisoles in sheep and goats might be relevant. Fig. 4 illustrates the findings and demonstrates that peak blood levels occur in goats very soon after oral drenching, fall rapidly and do not achieve concentrations comparable to those in sheep.
Hall et al. (1981), Kettle and White (1982), Gilham and Obendorf (1985) have now shared their concerns on the efficacy of anthelmintics currently used in goats with those of other veterinarians.
All of us must continue to insist that dose rates for goats and sheep should be evaluated independently.
Further we must ensure that metabolic profiles are obtained for cashmere, angora and dairy goats as a safe-guard against dangerous extrapolation across these types.
The goat industry is expanding and the cottage industry image is fading quickly. Demands will be put on us to produce control programmes and no matter how sound that programme is, its success will be limited by two main factors.
1. The presence of resistant parasites.
2. The survival of parasites because inappropriate dose rates have due to been applied or/an over-efficient metabolic process in goats which eliminates the anthelmintic too rapidly.
Whatever the cause the result will be the same and in time, factor 2 will produce factor 1.
It is not practically possible in all cases to isolate field strains of parasites in goats and passage through sheep to check if the parasite is truly resistant to the anthelmintics used.
We have to accept the lack of efficiency when it occurs and attempt to evaluate a dose rate at which a better kill is effected.
The use of Seponver in goats The Northern Tablelands is a H. contortus endemic area and the Wormkill programme relies on Seponver to control this major pathogen.
The programme was tested experimentally in sheep and it was shown that we could expect up to 12 weeks sustained activity by Seponver against H. contortus. The programme has been highly successful against Haemonchus so much so that the parasite is rarely seen nor its larvae cultured. It should be possible to reduce the number of Seponver treatments applied in future years.
Wormkill has been adopted widely for use in goats and we are faced with the circumstances of using an unregistered product in goats applied at sheep dose rates. Can we expect the same level of sustained activity from Seponver used in goats when it is applied at 12 weekly intervals at sheep dose rates or will dose rates have to be modified and/or the time interval between drenches shortened?
Table I gives some useful if limited data. We have two properties which are monitored routinely prior to each strategic drench time and on which sheep and angora goats are run. These properties, 1 and 2, afford direct comparisons between sheep and goats on Wormkill. The Haemonchus control is what we find typically throughout the Northern Tablelands for Wormkill sheep some 12 weeks post-Seponver treatments. That is, virtually nil egg counts for this endangered species. Properties 7 and 8, however, pose some questions and whilst the Haemonchus egg counts are still insignificant from an animal health and pasture contamination point of view, the larval differentiation % levels greater than 25 are worrying.
Are we experiencing from these limited results a major difference between dairy goats and the fibre goats with respect to drench clearance or was the weight estimation on properties 7 and 8 so inaccurate that an under-dosing situation had occurred.
I have attempted to persuade the manufacturers of Seponver to proceed with registration of this product for use in goats and elucidate dose rates. These pleas have not been successful to date.
It is however, an invaluable product for use in summer rainfall areas including the North Coast districts, Upper and Lower Hunter where haemonchosis is a real problem.
I would feel confident in advocating its use in goats at 3 monthly intervals, the more cautious veterinarians may prefer to use it at 8 weekly intervals.
The use of Seponver gives veterinarians an opportunity to control H. contortus effectively using a narrow-spectrum anthelmintic. Once this occurs the situation with regard to T. colubriformis and O. circumcincta is clarified. Success or failure from that point will be dependent on the presence and degree of anthelmintic resistance.
Conclusion
In the Norther Tablelands we have become inured to the effects of anthelmintic failure in sheep because of the high prevalence of resistant T. colubriformis and O. circumcincta. Dealing with anthelmintic resistance or failure due to the unique metabolic processes of the caprine is perhaps just a further extension of this basic problem. It is common place to recommend double dose rates of one or both action groups of broad-spectrum drenches to combat resistant parasites in sheep and goats.
That goats are intrinsically more susceptible to gastro-intestinal parasites than sheep with a poor development of age acquired immunity aggravated by the unknown factors in the use of anthelmintics in goats & this is not a scenario we would choose to work in.
However I believe that goats do have the ability to develop resistance to round worm infestations once they have experienced the parasites and therefore it should be possible to run them in higher rainfall areas and fit them into a strategy that has been found suitable for use in sheep. Whether this is Wormkill, Drenchplan or any other strategy using minimal applications of broad-spectrum drenches and narrow-spectrum drenches wherever possible.
It is nevertheless an absolute essential that producers institute a weighing procedure so that their judgement of live weights becomes more accurate and a more proper basis for deciding at what dose rates to give anthelmintics, whatever the times factor above sheep dose rates is being aimed for.
This can be illustrated by a case history of a New England property running cashmere goats primarily for blackberry control but with an increasing emphasis on upgrading programmes. In June 1984 a parasitological disaster occurred with deaths, particularly in his upgraded and more intensively run goats, due to T. colubriformis. Mean egg counts in the order of 4,328 epg and total worm counts showing 36,000 T. colubriformis. Suppressive monthly drenching with levamisole/morantel group anthelmintics had not stemmed the tide.
A faecal egg count reduction trial was conducted at a 4 day interval with the following results:
| Anthelmintic | Total mean eggs/gram faeces | % Reduction | |
|---|---|---|---|
| Day 0 | Day 4 | ||
| Systamex | 2084 | 120 | 94% |
| Nilverm | 2311 | 617 | 73% |
| Exhelm-E | 3949 | 2048 | 48% |
It was decided to put the goats on Wormkill using a third generation benzimidazole.
The sin of omission, in hindsight, was a failure to monitor the goats prior to each strategic drench time. Something I would advocate for all budding caprinolgists in the parasite spheres.
A minor hiccup occurred in mid-January 1985 the following year but was quickly corrected by using an extra broad-spectrum drench.
January 1986 was disastrous particularly in young stock run on sown pasture and no access to browse. Seponver and supposedly a sheep double dose of Systamex had been applied in November 1985 and the results are shown below at the time they were due for their next drench on the 1st February, 1986.
| Stock | Mean Strongyle epg | Larval Differentiation % | ||
|---|---|---|---|---|
| H. | T. | Ost. | ||
| 4-5 month old does | 2680 | 4 | 74 | 26 |
| Adult does | 2980 | - | 80 | 18 |
| 4-5 month old bucks | 680 | - | 67 | 33 |
| 2 weeks post a Systamex x 2 | ||||
This gives a reduction in faecal egg count of 74% 2 weeks after a supposed double dose of Systamex.
This situation was no worse than June 1984 after the suppressive use of levamisole/morantel groups but was still less than acceptable.
Further investigation is yet to be completed on this property by the Veterinary Inspector Armidale who has made an interesting observation. Weighing procedures have been instituted only to discover a gross under estimation and therefore the goats had not been getting a double dose of a benzimidazole at all.
On going work on this property continually reminds me of the dangers of overstocking goats and limiting access to browse.
It is in these situations of multiple drench resistance where we might be tempted to supplement our anthelmintic strategies with perhaps the use of low dose phenothiazine in blocks such as Wormolas.
Under field conditions the rate of voluntary intake of these medicated blocks is variable and this is probably the greatest disadvantage of the system. How do we ensure 5g of block/day/goat?
The desired advantages of using Wormolas blocks would be to significantly reduce the faecal egg output and infective larval production in both susceptible and resistant strains of T. colubriformis and O. circumcincta.
Kelly et al. demonstrated a highly significant effect of low-dose phenothiazine in suppressing worm egg production and inhibiting larval hatching in strains of H. contortus, T. colubriformis and O. circumcincta known to be susceptible to benzimidazoles, levamisoles and morantel tartrate in sheep. This would effectively reduce pasture contamination. A 5 gram daily dose rate of the blocks is needed whilst this does not effect established worm number and complementary therapy is needed.
There is, however, a reduced efficiency of Wormolas treatment against parasite strains which are resistant to benzimidazoles, levamisoles and morantel. The common denominator being the benzimidazole resistance suggesting a cross linked phenothiazine/benzimidazole factor.
Viability and infectivity of larvae and so establishment rates are higher in resistant strains compared to susceptible strains. A fine selection for survival.
We may therefore, in our attempts to control parasites in goats confound the whole anthelmintic therapy scene by using phenothiazine blocks and exacerbate the benzimidazole inefficiency.
Perhaps when all is virtually lost anyway there can be little further harm done by using the blocks in an attempt to reduce pasture contamination along with the strategic use of double, treble doses of anthelmintics - singularly or in conjunction not forgetting that Rametin - H, whilst potentially toxic, can give a breathing space away from broad-spectrum drenches to control T. colubriformis.
Veterinarians in the D.P.I. Queensland (Personal Comm). are using Rametin - H as the last ditch stand. Owners are acquainted with the risk and accept it.
They will die if we do nothing! Another possible course of action is the application of a broad spectrum anthelmintic twice - at day O and day 5. Hopefully to maintain blood levels and effect a better kill.
None of these desperation methods solves the basic dilemma.
Accountability to the goat industry and subsequently the sheep industry is in question in terms of the development of resistant strains of parasites due to the application of inappropriate dose rates or preparations to goats.
Enforcing requirements that registration of an anthelmintic for use in goats must include the clarification of dose rates might ensure that products are never registered.
Unfortunately the goat industry has very little muscle at the present time and the order of priority is against work done in this species. In the Northern Tablelands we feel the pressure of this expanding industry and its significance to parasite control in sheep. We must continue to pressure for work to be done on the dose rates and duration of action of anthelmintics in caprines.
A small glimmer of hope. Avermectin for sheep and goats may well be with us in two years time.
We are reassured by its manufacturers that because of the stringent regulatory requirements in European countries that appropriate dose rates for goats have been ascertained and clear recommendations will be made.
Acknowledgements
To Steve Love, Veterinary Inspector Armidale, whose field monitoring data is included in this paper together with the writer's. Interchange of information is invaluable.
To Mike Emms, Veterinary Inspector Denman/Singleton for his pioneer work in parasite control in goats in the lower Hunter, where he is applying a modified Wormkill programme. He has instituted regular monitoring and weighing procedures.
The results given below are extremely encouraging on this dairy herd.
| Date | epg | Larval Differentiation % | |||
|---|---|---|---|---|---|
| H. | T. | Ost. | Oe. | ||
| 12.7.85 pre 'Wormkill' | 3290 | 26 | 54 | 17 | 3 |
| 4.8.85 Seponver and Nilverm | |||||
| 9.8.85 | 177 | 19 | 69 | 12 | 0 |
| 4.10.85 | 607 | - | 55 | 22 | 23 |
| 6.10.85 Drench with Seponver and Nilverm | |||||
| 11.10.85 | 80 | - | 51 | 18 | 31 |
The owner is delighted with the results.
REFERENCES
- L.F. Jambre and W.M. Royal, (1976) 'A Comparison of Worm Burdens in Grazing Merino Sheep and Angora Goats' A.V.J. Vol 52 No. 4 P. 181-183
- McCulloch, B. and Kasimbala, S. (1968) British Veterinary Journal 124:177
- LeRich, et al. (1973) J. Helminth. 57:251
- Silangwa S.M. & Todd, A.C. (1964) J. Parasit. 50:278
- L.F. Le Jambre (1984) 'Stocking Rate Effects on the Worm Burdens of Angora Goats and Merino Sheep' 29.4.84.
- L.F. Le Jambre (1977) 'Ostertagia ostertagi Infections in Angora Goats' Veterinary Parasit. 4 (1978)
- Southcott, W.H. & Barger, I.A. (1975) Int. J. Parisol. 5:45-48
- Kettle et al. (1983) New Zealand Veterinary Journal 31:139-143
- Kettle P.R. & White, D.A. (1982) 'Anthelmintics and their use against Nematodes in Goats' N.S.V.A. Seminar on Dairy Goat Husbandry Whangarei, December 1982
- Gillham R.J. & Obendorf, D.L. (1985) 'Therapeutic Failure of Levamisole in Goats' - received for publication 1985, authors agreed on use of paper.
- Hall, C.A. et al. (1981) Res. Vet. Sci. 31:116-119
- S.W. Walkden-Brown (1985) 'Small Ruminant Production Systems in Fiji - Problems and Prospects'. Aciar Workshop on the Epidemiology and Control of Gastro-Intestinal Nematodes, in Armidale, N.S.W. 19-21st November, 1985
- G. de Chaneet & R. Lewis (1973) 'The use of Molasses Blocks Medicated with Phenothiazine to Control Nematodes in Sheep in W.A.' Australian Veterinary Journal, Vol. 49, March 1973
- J.D. Kelly, H.V. Whitlock, C.J. Porter, D. Griffin, I.C.A. Martin 'Anthelmintic Efficacy of Low-dose Phenothiazine Against strains of Sheep Nematodes Susceptible or Resistant to Thiabendazole, Levamisole and Morantel Tartrate: Efficiency against Sequentially Administered Infections'. Research in Vet. Science 1981, 30, 170-174
- J.D. Kelly, H.V. Whitlock, M. Gunawan, D. Griffin, C.J. Porter & I.C.A. Martin. 'Anthelmintic Efficacy of Low-dose Phenothiazine against Strains of Sheep Nematodes Susceptible or Resistant to Thiabendazole, Levamisole and Morantel Tartrate: Effect on Patent Infections'. Research in Vet. Science 1981, 30, 161-169
- T.E. Gibson, 'Veterinary Anthelmintic Medication' Third Edition - Technical Communication. Page 211-218
FIG. I
| ADULT SHEEP (& HOGGETS) | LAMBS | |||
|---|---|---|---|---|
| Date | Seponver | Broad-spectrum drench | Seponver | Broad-spectrum drench |
| 1st August | * plus | * | ||
| 1st November | * plus | * | * plus | * |
| 1st February | * | * plus | * | |
| 1st April | * | |||
* = Manufacturer's recommended dose calculated for the heaviest sheep in the group.
FIGURE 2.
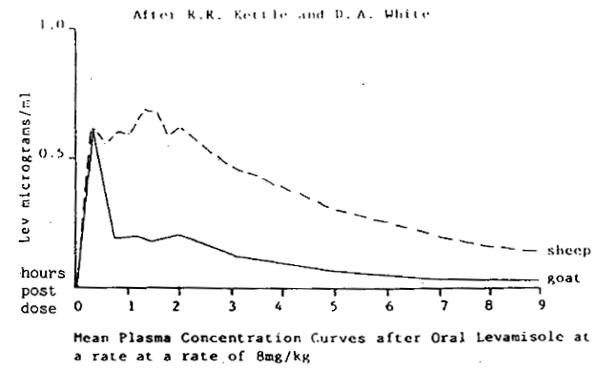
FIGURE 3.

FIGURE 4. Mean plasma concentration curves of levamisole after oral administration to goats at 7.1 mg/kg (◻) and sheep at 7.5mg/kg (●) (sheep data after Bogan et al. 1982).
Levamisole (g/ml) Plasma os 20 40 60 80 120 150 240300 Time (min)

TABLE 1.
Results of monitoring goats which are on the Workill programme. As with standard monitoring procedures in the faecal samples are collected just prior to each strategic drenching time: Ten samples per group.
| Animal Group | Mean Strongyle epg | Larval differentiation % | |||
|---|---|---|---|---|---|
| Period at least 10/12 weeks after the last drench unless otherwise indicated | *H. | *T. | *Ost. | ||
| 1. | Angora Goats | 808 | 1 | 82 | 12 |
| Sheep | 1345 | 1 | 84 | 7 | |
| Angora Goats | 380 | - | 96 | 4 | |
| Sheep | 220 | - | 97 | 3 | |
| 7 Days Post Drenching | |||||
| Goats | 318 | 1 | 82 | 17 | |
| Sheep | 488 | - | 89 | 9 | |
| Goats | 264 | - | 94 | 6 | |
| 7 Weeks Post Seponver + B'Spec. | **888 | - | 97 | 3 | |
| ** Nutritionally stressed group | |||||
| 2. | Angora Goats | 190 | 1 | 86 | 13 |
| Sheep | 127 | - | 58 | 41 | |
| Goats (includes kids) | 806 | - | 79 | 21 | |
| Sheep | 160 | - | 97 | 2 | |
| Goats | 556 | - | 85 | 15 | |
| Sheep | 248 | 1 | 84 | 14 | |
| 3. | Angora Goats | 400 | 1 | 81 | 18 |
| Goats | 216 | 1 | 80 | 19 | |
| Goats (kids) | 514 | - | 82 | 18 | |
| 4. | Cashmere Goats | 2830 | - | 77 | 22 |
| 5. | Angora Goats | 177 | 1 | 67 | 30 |
| 451 | - | 63 | 37 | ||
| 240 | 5 | 47 | 42 | ||
| 6. | Angora Goats | 1380 | - | 99 | 1 |
| 7. | Dairy Goats | 287 | 25 | 25 | 44 |
| Known multiple drench resistance | 143 | 3 | 42 | 55 | |
| 552 | 51 | 12 | 37 | ||
| 451 | 21 | 36 | 42 | ||
| 8. | Mixed Goats (fibre/dairy) | 60 | 77 | 22 | 1 |
| 192 | 74 | 20 | 1 | ||
H. H. contortus
T. T. colubriformis
Ost. O. circumcincta