
ARCHIVE FILE
This article was published in 1985
See the original document
Residues in Cattle on Exposure to Land Contaminated With Heptachlor
I.R. Harradine, B.V.Sc., Veterinary Inspector, GRAFTON and K.W. McDougall, Laboratory, Board of Tick Control, LISMORE
SUMMARY
Three groups of four steers were grazed on a former potato paddock which had been treated twice previously with heptachlor at the rate for the control of African Black Beetle. Soil residues in the paddock varied from a total of 0.42 mg/kg heptachlor at the beginning of the trial to 0.31 mg/kg after 16 months. Residues decreased only slightly in the soil down to a depth of 300mm. Pasture residues were <0.02 mg/kg total heptachlor (wet weight).
No heptachlor residues were detected in fat but its metabolite, heptachlor epoxide, increased progressively over 19 months of exposure and reached maximum levels of 0.72 mg/kg. In 4 steers removed after 14 weeks exposure the heptachlor epoxide concentrations continued to increase from a mean of 0.24 mg/kg to a mean of 0.34 mg/kg after a further 4 weeks. Concentrations then fell progressively with a half-life of 11 weeks in the body fat.
There appeared to be a relationship between pasture length and the intake of heptachlor. It is suggested that the latter occurs through the ingestion of contaminated soil which, in turn, increases as the length of pasture decreases.
INTRODUCTION
The organochlorine insecticide (1,4,5,6,7,8,8 heptachloro-3a,4,7,7a tetrahydro-4, 7-methanoindene) has been used extensively in Australia for the control of many soil pests. In particular, heptachlor has been used to control African Black Beetle in potato horticulture. This posed a problem for farmers who wished to combine potato growing and cattle grazing. Gilbert and Lewis (1982) showed that cows produced residues in the milk fat which exceeded the Maximum Residue Limit (MRL) when continuously exposed to contaminated land. Solly (1967) and Solly et al. (1968) showed that low levels of heptachlor epoxide resulted in the body fat of sheep grazed on treated land or pasture after various withholding periods of 5-45 weeks.
The present trial was undertaken to determine the rate of build-up of residues in cattle when exposed to land treated with heptachlor and the rate of decline on removal from such contaminated areas.
MATERIALS & METHODS
SOIL & PASTURE
Heptachlor was incorporated into the soil of a 7 ha paddock at Clouds Creek, 70 kilometres west of Grafton, N.S.W. The paddock was fertilised and treated with heptachlor at the recommended rate for African Black Beetle of 1.1 kg/ha of 25% active ingredient in August 1979 and again in August 1980.
The paddock was divided into 20 equal area plots and, using a 75mm long x 20mm diameter soil core sampler, 10 cores were collected randomly from each plot. Samples were collected in July and October 1982 and again in June and November 1983. For each plot cores were bulked together and mixed thoroughly before analysis.
Pasture was collected in July 1982 using a hand held reaper. Each plot had pasture cut at random to give a total of approximately 0.5 kg per plot. Pastures were stored frozen at [-] 20C until analysed. The length of the pasture was measured at ten sites throughout the paddock each month.
In April 1982, two groups each of four Hereford steers, 20 months old and in good condition, were placed on the contaminated area. Fat samples were taken prior to, and 10 weeks after, their introduction. After 10 weeks one group of four steers was withdrawn to the Grafton Agricultural Research & Advisory Station and grazed in pasture on which heptachlor had never been applied. At the same time a third group of four steers, 22 months old, were fat sampled and introduced to the Clouds Creek property. Further samples were taken from this group every 2 weeks for an eight week period and then all twelve cattle were fat sampled at approximately one month intervals. The cattle were also weighed at intervals.
The cattle were allowed to graze on sown rye grass (Lolium perenne) pastures covering the affected area and no supplementary feeding was given during the duration of the experiment. Towards the end of the trial pastures had reverted to natural kikuyu (Pennisetum clandestinum) and paspalum (Paspalum dilitatum). Water was available from a creek running through the lower part of the property.
The biopsy technique of Saville et al. (1972) was employed, except that 2% xylocaine was used as an anaesthetic instead of dry ice. All samples were frozen before transporting to the laboratory.
CHEMICAL ANALYSIS
Soil and Pasture - Pasture samples were chopped, mixed, blended with acetone, filtered and evaporated to dryness. The extract was then passed through a florisil column and evaluated for heptachlor and its metabolite, heptachlor epoxide by gas chromatograph (GC). This method is based on that of Milton et al. (1975).
Soil samples were mixed and a subsample extracted in a Soxhlet apparatus for 2 hours with acetone. The extract was then evaporated to dryness and treated as for pasture. Recoveries of heptachlor and heptachlor epoxide were greater than 95% from both soil and pasture. Body fat samples were first rendered with heat to produce pure fat. The residues were then extracted using the assisted distillation technique of Heath and Black (1979) with modifications by McDougall (1980). Recoveries of heptachlor and heptachlor epoxide were 90±4%.
Residues were determined on a Varian 2100 GC equipped with electron capture detector. The glass 2m column had an internal diameter of 2mm and was packed with 3.75% OV210/2% SE30 on Gas Chrom Q (80-100 mesh). Column temperature was 205°C.
RESULTS CATTLE
Mean heptachlor epoxide concentrations found in fat of each group of steers are shown in Fig. 1. No heptachlor residues were found in the steers at the start of the period of exposure. In the group 2 steers, concentrations had risen to an average of 0.24 mg/kg when they were removed from contaminated pastures in August 1982. Concentrations then continued to rise to a peak of 0.34 mg/kg in September 1982. Thereafter they declined and reached background levels in June 1983, 9 months later. This gave a calculated half life of 11 weeks for heptachlor epoxide in body fat of steers.
Group 1, introduced in April 1982, and group 3, introduced in August 1982, reached average levels of about 0.3 mg/kg heptachlor epoxide by September 1982. Thereafter levels remained reasonably constant until about February 1983 after which they increased steadily to reach levels of 0.68 mg/kg in September 1983 which were maintained generally through to December 1983. During the period October 1982 to November 1983 the cattle increased in mean liveweight from 312 kg to 475 kg.
SOIL & PASTURE
Soil concentrations of heptachlor and heptachlor epoxide are shown in Table 1. In July 1982 heptachlor was the major component at a concentration of 0.274 mg/kg while in November 1983 heptachlor epoxide was the major component (0.177 mg/kg).
In a further investigation of soil residues in this paddock 4 samples were taken and each one divided so that soil residue profile could be studied. This study showed that soil residues decreased only slightly over the depth of 300mm examined. A sample of soil taken from top soil deposits in the creek following heavy rain returned results of 0.90 mg/kg heptachlor epoxide and 0.05 mg/kg heptachlor.
Residue levels in undried pasture taken in July 1982 were <0.02 mg/kg heptachlor and heptachlor epoxide. Due to the decreasing length of the pasture no further samples were able to be taken. The pasture was initially 255mm long and over the period of the trial decreased to 30mm (Fig. 1).
DISCUSSION
It appears from this study that cattle will exceed the Maximum Residue Limit (MRL) of 0.2 mg/kg heptachlor and its epoxide when grazed on soil treated with heptachlor. In fact it took less than one month for the steers in the third group to exceed the MRL after commencement of grazing. This would mean that even with occasional exposure the MRL would probably be exceeded.
There appeared to be a relationship between the length of pasture and the intake of heptachlor. Thus the concentrations in fat increased progressively as the length of pasture decreased to October, 1982 remained fairly constant when pasture length and availability was greater, to February, 1983 (Fig. 1). Thereafter the concentrations increased progressively as pasture became shorter due to drought conditions and high stocking rate.
Little heptachlor was found in pasture material but considerable amounts were present in the soil. This suggests that the concentration of heptachlor in the animals was related to the ingestion of soil and that the latter increased as pasture length decreased and the cattle grazed closer to the ground. The gradual absorption from ingested soil could also explain the increase in concentrations which were detected in the steers of Group 2 after they were removed from the contaminated paddock.
Reasons for the variable soil concentrations are unknown. They could be due to extreme variability in the applications resulting in 'hot spots' which are not always sampled. The overall rate of decline in a 16 month period is almost negligible which supports published data on the half. life of heptachlor (7-12 years) (Menzie 1972). This means that for many years following application of heptachlor, residues persist in the soil and present a means of contamination for any grazing animal allowed access to it.
The presence of soil residues down to 300mm is as expected. The process of harvesting potatoes results in extensive mixing of soil to a depth of 250-300mm. The presence of heptachlor epoxide in the soil deposits in the creek bed is a significant finding when considering possible contamination of cattle grazing lands below such cultivated areas.
All these facts preclude the option of grazing cattle on heptachlor treated soil. The mixing of horticulture with animal production in this particular case is not feasible if residues of heptachlor in the body fat are to be kept below the MRL. It is obvious that where stocking rate affects pasture length then this in turn affects body fat residues.
ACKNOWLEDGEMENTS
We wish to thank A. Heath and J. O'Connor and their staff for analysis of samples, C. Thompson, M. Davies, R. Warren and J. Sedwell for assistance in collecting samples and the Pastures Protection Board, Grafton for the use of clouds Creek Travelling Stock Reserve and provision of fencing.
REFERENCES
Gilbert, W.S. and Lewis, C.E. (1982) - Australian Journal of Experimental Agriculture Anim. Husb. 22:106
Heath, A.B. and Black, R.R. (1979) - J. Assoc. Off. Anal. Chem. 62:757
Luke, M.A., Froberg, J.E. and Masumoto, H.T. (1975) - J. Assoc. Off. Anal. Chem. 58:1020
McDougall, K.W. (1980) - J. Assoc. Off. Anal, Chem. 63:1355
Menzie, C.M. (1972) - Annu. Rev. Entomol. 17:199
Saville, D.G., Gilbert, W.S. and Wright, J.M. (1972) - Australian Journal of Experimental Agriculture Anim. Husb. 12:460
Solly, S.R.B. (1967) - Proc. Weed Pest Cont. Conf. 1967:167
Solly, S.R.B., Harrison, D.L. & Shanks, V. (1968) - N.Z. J. Agric. Res. 11:371.
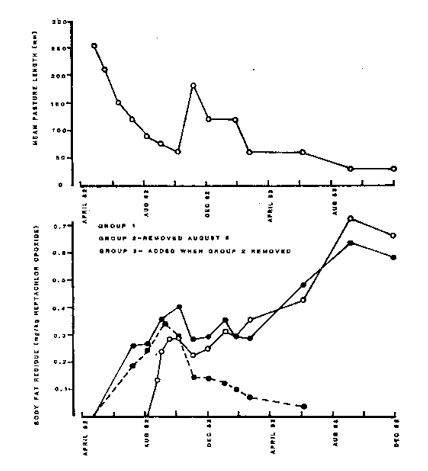
Table 1:
Mean heptachlor residues in soil samples taken from an ex-potato field at Clouds Creek NSW
| SAMPLING DATE | MEAN RESIDUE ± SE (mg/kg)* | ||
|---|---|---|---|
| HEPTACHLOR | HEPTACHLOR EPOXIDE | ||
| July 1982 | .274 ± .075 | .148 ± .019 | |
| October 1982 | .054 ± .024 | .033 ± .015 | |
| June 1983 | .078 ± .014 | .108 ± .015 | |
| November 1983 | .138 ± .063 | .177 ± .033 | |
* Each result is the mean of 20 samples.