
CASE NOTES
Mycotoxicosis in a Merino flock
Megan Davies, District Veterinarian, Narrabri / Walgett
Posted Flock & Herd February 2015
Introduction
Mycotoxins are secondary mould metabolites produced by certain species of fungi, including Aspergillus and Fusarium spp. They are commonly found in grain, hay and silage, and occur as a result of fungal contamination of the feed during growth, harvest or storage. Ruminants are generally able to cope with higher levels of mycotoxins in feed than monogastrics, however impacts on production can still be significant, and large numbers of losses can eventuate. There is no specific antidote for mycotoxicity, and immediate removal of the contaminated feed is the single most important step in avoiding further losses.
History
A mixed group of 600 merino ewes, lambs and wethers were grazing native pasture in North Western NSW in October 2014. The owner noted two had died on Monday, followed by 20 more on Tuesday, when the district veterinarian was called to investigate. By Wednesday approximately 50 sheep were dead, with approximately 10 more showing neurological signs including tremors, seizuring, paddling of limbs and blindness. 80 sheep had died before the episode had run its course.
The sheep had been vaccinated with 5-in-1 at lamb marking approximately two months earlier. They had not been recently drenched. They were grazing largely native pastures, with Fuchsia Bush (Eremophila maculata - a cyanogenetic glycoside) present in moderate amounts, with evidence that the sheep had been grazing these plants. The flock had been yarded one week prior and the lambs had been jetted with a lice treatment and held overnight. The property was in drought, with high spring temperatures recorded the previous week (40°C for three to four days straight). There had been no recent rainfall.
The owner was supplementing the sheep with good quality barley hay sourced from a regular supplier. He had also recently cleaned out a silo and spread the old grain from the base of the silo around the paddock in piles that the sheep had been consuming. Following an initial phone consultation, the remaining sheep were removed from the paddock in which the deaths occurred. Deaths continued in low numbers for two to three days after removal from the paddock.

Figure 1: Sheep found dead in paddock

Figure 2: Grain from Silo in piles throughout paddock

Figure 3: Grain from cleaned-out silo, fed out to sheep in paddock
Clinical examination
Several live affected sheep were examined. All were ataxic, with marked weakness of fore and hind limbs, some unable to stand, with knuckling of the fetlocks. Some were able to stand when lifted, but staggered and circled before falling down again.
Several were in lateral recumbency with all four limbs paddling uncontrollably. All affected animals had significant muscle tremors, and all except one were blind. Two animals were having grand mal seizures, with one being euthanased (Ewe 1) and the other dying shortly after examination (Ewe 2).
Multiple dead animals were observed, many along fence lines and under trees. All showed evidence of paddling of limbs prior to death. Several were tangled up in tree branches. Two were observed to have frothy bloody nasal discharges. One recently deceased ewe was autopsied (Ewe 3). An anthrax immunochromotographic test performed in the field on Ewe 3 was negative.

Figure 4: Dead sheep with evidence of paddling of limbs prior to death
Post-mortem findings
Three full post-mortem examinations were performed on adult ewes 1, 2 and 3. All showed enlarged pale tan coloured livers with nutmeg patterns throughout the parenchyma. All had distended gall bladders with patent bile ducts. All showed dark purple to red kidneys with a red striped pattern throughout the cortex. Two ewes (1 & 2) also had haemorrhages within the myocardium of the ventricles. All three animals had a layer of dirt covering the mucosa of the trachea extending into the bronchi. Samples were submitted under the Transmissible Spongiform encephalopathy (TSE) program.
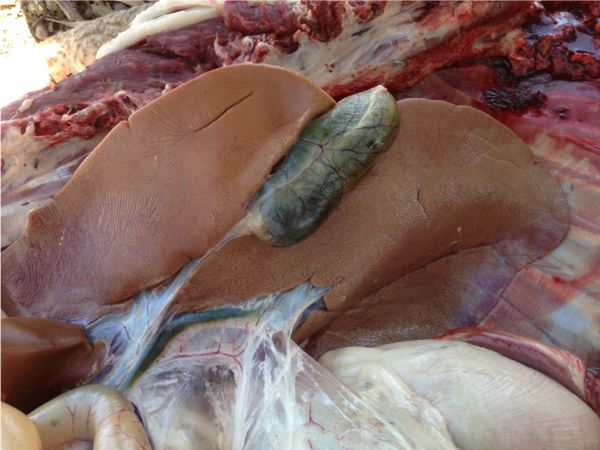

Figures 5 & 6: The Liver of Ewe 1 shortly after euthanasia

Figure 7: Damage to myocardium evident in Ewe 1

Figure 8: Kidney of Ewe 1 showing striped pattern through the cortex
Pathology results
Initial pathology results from the State Veterinary Diagnostic Laboratory at Menangle showed "No significant histopathological brain lesions suggesting cause of neurological signs in these ewes; no cerebrocortical grey matter lesions suggestive of polioencephalomalacia; no evidence of hepatic encephalopathy."
The liver of Ewe 1 showed "Portal triads show moderate oval cell hyperplasia and proliferation; occasional mitotic figure noted. Mild hepatic lipidosis". Ewe 2 showed "Mild fatty hepatocyte degeneration. Occasional mild portal lymphocytic infiltrate." The hepatic lipidosis did not appear severe enough to cause hepatic failure, but did suggest a recent toxic insult to the liver. A mycotoxin was suggested as a possible cause.
Blood results from ewes 1 & 2 showed that "both sheep have elevated AST and CK consistent with muscle damage probably due to recumbency, tremors and convulsions". Both animals also had mildly elevated ammonia levels, however these were thought to be due to the delay in testing, rather than due to urea poisoning.
Ewe 1 also showed moderately elevated GGT (120 U/L (0-55)) and GLDH levels (274U/L (0-30)), further consistent with a liver insult.
A grain sample was submitted for mycotoxin screening. The results showed the feed sample submitted contained Aflatoxin B1 at 140ppb (0.14mg/kg) and Deoxynivalenol (DON) at 670ppb - these levels will vary within the grain storage.
Diagnosis
A diagnosis of aflatoxicosis was considered most likely in this case, owing to the sudden onset of the deaths, and the fact that they stopped soon after the animals were removed from the source of the aflatoxins. Deoxynivalenol (DON) may also have contributed to the deaths by potentiating the activity of the aflatoxins. The amount of fuchsia bush present in the paddock was not thought to be significant enough in this instance to account for the number of deaths, nor did the clinical and pathological evidence fit with a mass incidence of native fuchsia toxicity.
Discussion
Mycotoxicosis is well reported in monogastrics and dairy cattle; however reports of affected sheep are limited. There are many difficulties in obtaining a definitive diagnosis of mycotoxicity, due to the frequent lack of visible symptoms, difficulty of gaining a representative sample of feed (Bryden et al. 1980), proving ingestion by the affected animals, and screening for over one hundred possible mycotoxin culprits. Interpretation of laboratory results is complicated by dry matter percentages of feed, units used in reporting, how much of the ration is made up of the contaminated feed, and whether mycotoxins are present in other ration ingredients (Adams et al. 2014).
Diagnosis is further confounded by the fact that the effects of mycotoxins are accumulative over time, and, on farm, the presence of more than one mycotoxin may potentiate or increase the harmful effects (Adams et al., 2014). Environmental factors, exposure levels, age, health and nutritional status can also influence the toxicity and its subsequent clinical presentation (Saini & Kaur, 2012 and Cassel et al. 1989).
In this instance, two mycotoxins were detected, however only Aflatoxin has been reported to cause marked neurological symptoms. Recent research indicates that the presence of other mycotoxins, in this case Deoxynivalenol, could exacerbate symptoms and contribute significantly to the disease process (Adams et al., 2014).
AFLATOXINS
Aflatoxins are secondary mould metabolites produced by Aspergillus flavus and Aspergillus parasiticus species (Cassel et al. 1989). Aflatoxin B1 is the most toxic of this group of compounds and no animal species is resistant to its acute toxic effects, however there is variability in susceptibility between species (Saini and Kaur, 2012). Potentially harmful levels for cattle are reported as 20-300ppb (Adams et al. 2014), so the level of 140ppb found in this case would very likely have been harmful to the sheep.
Aspergillus are common fungi often found on stored foods, particularly peanuts and maize, but also seen in grains including wheat and barley (Bryden et al. 1980) and grain byproducts (Cassel et al., 1989).The infestation may occur during growth of the plant, or after harvest, before the grains have dried out sufficiently to prevent mould growth. Hot, humid conditions favour the development of aflatoxins (Adams et al., 2014), with optimal conditions for growth of the fungus being relative humidity greater than 90% and temperatures of 30-40°C (Seawright, 1989). Farm silos are reported to be a major site of toxin production in mould-contaminated feeds (Bryden et al. 1980) as storing feedstuffs at moisture levels beyond recommended ranges or in poor storage units can encourage mould growth (Adams et al. 2014). Once spores begin to grow, they produce heat and moisture as a by-product, triggering the growth of more spores in a snowball effect (Wilcox, 2014).
Sheep are reported to be relatively resistant to the effects of aflatoxins when compared with other species (Plant, 1992). The major target for the toxicity of Aflatoxins is the liver. Acutely affected animals may die suddenly, without clinical signs. If symptomatic, sheep may show tremors, lameness, ataxia and recumbency (Radostits, 1994). In cattle, reported clinical signs include blindness, walking in circles, frothing at the mouth and knuckling at the fetlocks. Less acutely affected animals show anorexia, depression, ataxia, dyspnoea and anaemia (Seawright, 1989).
Chronic aflatoxicosis causes reduced food conversion efficiency, with weight loss, rough coat, anaemia and jaundice being common clinical signs (Seawright, 1989).
Necropsy findings relate to acute or chronic liver damage, depending on the level of aflatoxin present and the duration of feeding (Cassel et al. 1989). Aflatoxins produce necrosis of liver cells, damage to mitochondria and proliferation of bile ducts. The liver is usually pale tan or yellow, with hepatic fibrosis and oedema of the gall bladder (Cassel et al. 1989). The toxins also cause centrilobular necrosis and marked fatty infiltration of the liver (Seawright, 1989). Degenerative changes in the nervous tissue may also be seen (Radostits et al. 1994), and vascular haemorrhages have been reported (Wilcox, 2014).
There is significant variability in the reported levels of Aflatoxins required to cause death in sheep. Radostits et al. (1994) reports that a dose rate of 4mg/kg causes death at 15-18 hours due to acute hepatic insufficiency, with heavy mortalities having occurred. Bryden et al. (1980) report clinical signs in cattle at levels as low as 0.1mg/kg. Deaths may continue for several weeks, with a few animals dying as late as 5 months later (Seawright, 1989).
Deoxynivalenol
Deoxynivalenol (DON) or "vomitoxin" is a group B Trichothecene produced by the fungi Fusarium spp. and has been reported to cause anorexia and staggering in dairy cattle, and vomiting and feed refusal in pigs (Seawright, 1989). Confirmed intoxications caused by this compound are uncommon in Australia, but well reported in the United States. Potentially harmful levels for cattle are reported as 2.5-6.0ppm, but levels as low as 0.6ppm can be harmful to pigs (Adams et al. 2014). The level of 670ppb (0.67ppm) in this feed therefore has the potential to be harmful, but on its own may not have been significant enough to cause clinical symptoms.
Fusarium is a common pathogen of wheat, and if it is detected, it is not recommended to feed the grain to livestock (P.Davies, pers com, 2014). Fusarium and its metabolites including DON are stable for long periods, and as a result their concentrations generally magnify during feed processing and storage. Monogastics are more severely affected by the toxins, with feed refusal occurring at very low concentrations within the feed (Seawright, 1989) however ruminants can also suffer from anorexia, diarrhea, haemorrhage and nervous disturbances.
Treatment and prevention
There is no specific antidote for mycotoxicity, and immediate removal of the contaminated feed is the single most important step in avoiding further losses. Full recovery following acute aflatoxicosis is rare due to the damage to the liver and other vital organs (Wilcox, 2014).
The most effective control strategy is prevention of fungal infection and toxin production in the field and in storage. Storage of grain at less than 13-14% moisture and hay and straw at less than 20% moisture is important in preventing mycotoxin production (Adams et al. 2014). There is no specific treatment for DON toxicity, but clinical signs of feed refusal usually disappear within a week of removal from the contaminated feed.
Most mycotoxins and Aflatoxin in particular, are difficult to remove from feedstuffs (Wilcox, 2014). Heat processing and ensiling do not destroy mycotoxins (Adams et al. 2014). Time and environmental exposure have limited effect on decreasing fungal levels in feed when spread out in the paddock, and even multiple large rain events have limited ability to reduce mycotoxin levels (P. Davies, Pers com, 2014).
Producer education and awareness of risks is an important tool in preventing losses due to mycotoxicity. This is particularly important during dry seasons and drought as feed becomes scarce and producers buy and feed out feedstuffs that may come from questionable sources or have been stored inappropriately.
References
- Adams RS, Kephart KB, Ishler VA, Hutchinson LJ & Roth GW (2014) Mold and Mycotoxin problems in livestock feeding. Department of Dairy and Animal science, The Pennsylvania State University, Pennsylvania
- Bryden WL, Lloyd AB & Cumming RB (1980) Aflatoxin contamination of Australian Animal Feeds and Suspected Cases of Mycotoxicosis Australian Veterinary Journal 56:176-180
- Cassel EK, Barao SM & Carmel DK (1989) Aflatoxicosis and Ruminants. University of Maryland, College Park, Maryland
- Davies PAB (2014) Personal communication Plant Pathologist (Wheat), Plant Breeding Institute, University of Sydney, Narrabri
- Finnie JW, Windsor PA & Kessell AE (2011) Neurological Diseases of Ruminant Livestock in Australia. II: Toxic Disorders and Nutritional Deficiencies Australian Veterinary Journal 89(7):247-253
- McBarron EJ (1983) Poisonous Plants, Handbook for Farmers and Graziers. Department of Agriculture New South Wales, Inkata Press, Melbourne
- Plant J (1992) The TG Hungerford Vade Mecum Series for Domestic Animals Series B Number 15 Diagnosis of Diseases of Sheep. University of Sydney Post Graduate Foundation in Veterinary Science, Sydney
- Saini SS & Kaur A (2012) Aflatoxin B1: Toxicity, characteristics and analysis: Mini Review Global Advanced Research Journal of Chemistry and Material Science 1(4):63-070
- Seawright AA (1989) Animal Health in Australia Volume 2 (Second Edition) Chemical and Plant Poisons. Bureau of Rural Resources, Department of Primary Industries and Energy. Canberra
- Radostits OM, Blood DC & Gay CC (1994) Veterinary Medicine; A textbook of the diseases of cattle, sheep, pigs, goats and horses. Bailliere Tindal, London
- Wilcox RA (2014) Moldy Feedstuffs and Potential Toxins. Cooperative Extension Service - Great Plains States. Boletim tecnico No. 39 www.microtoxinas.com.br Accessed 27/11/14